 French manufacturer BlueSom first launched BluePro® in Europe in 2014 and then received FDA clearance in December 2016 for the treatment of snoring and mild to moderate OSA. After launching in USA at AADSM/Sleep 2017 and receiving an encouraging response from US dentists, this same-day temporary appliance is now available nationwide.
French manufacturer BlueSom first launched BluePro® in Europe in 2014 and then received FDA clearance in December 2016 for the treatment of snoring and mild to moderate OSA. After launching in USA at AADSM/Sleep 2017 and receiving an encouraging response from US dentists, this same-day temporary appliance is now available nationwide.
Place in therapy
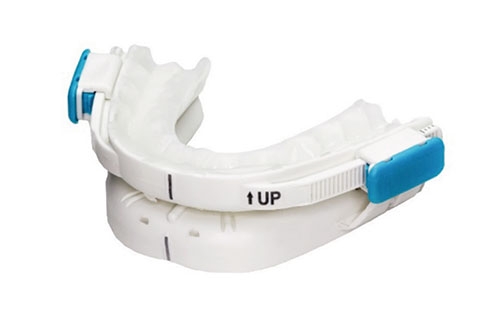
BluePro is prefabricated, adjustable and thermoplastic; a category increasingly termed ‘PAT-MAD’ in the literature. It is fitted chair-side by the dentist within a few minutes by immersing the two splints in boiling water, molding them directly on the arches, assembling to form a monobloc, protruding as tolerated to a comfortable starting position and then locking in position for patients to begin same-day treatment.
It plays a role comparable to other temporary appliances i.e. professional same-day transitional treatment, a way to affordably trial the patient’s response to mandibular advancement and a way to find an optimal level of titration comfortably through incremental adjustment. Indeed BluePro has demonstrated its suitability to this task in published studies and in European clinical practice. However a closer look at the design features and evidence reveals wider opportunities for use beyond the traditional role of a temp.
Retention
Professor Marc Braem evaluated this appliance in 2014 with a mechanical study to assess its retentive capability1. When molded to the teeth, the splints are designed to guide the pre-lined thermoplastic into the interdental spaces, creating a precise and durable impression. Professor Braem found the resulting mean force of retention to be around 26N, sufficient “to resist initial jaw opening as well as full mouth opening forces” over the equivalent of 1 year’s use.
Loss of retention is therefore rarely a problem for this device. It is more likely that patients will feel some tightness or dental discomfort, but this can be addressed during fitting by removing interdental material from the splint with a sharp knife.
Efficacy
Two independent studies, first a pilot in 24 patients2 and then a larger comparative study3 have confirmed the efficacy of BluePro in treating OSA. The second study analysed treatment results for two patient groups over six months; one group wearing BluePro (n = 86) and one group wearing custom devices AMO® or SomnoDent® (n = 72).
In their analysis, after adjusting for differences in the groups at baseline, the investigators observed no statistically significant difference between the groups in:
- Primary outcome measures of sleep- disordered breathing symptom relief (AHI, AI, ODI, T90);
- Mean measures of self-reported treatment satisfaction and compliance and;
- Overall side-effect scores calculated from self-reported measures (despite reports of greater dental discomfort in the BluePro group).
Of course BlueSom does not claim this to be evidence that the device can replace custom-made devices. However, the data shows that BluePro can provide equivalent efficacy to custom devices in suitable patients over a period of 6 months.
Wider role
In USA this device will be used mainly as a trial or transitional appliance and its durability allows it to be kept as a robust spare after the custom device is delivered. In Europe it has been frequently used as a pragmatic therapy device in settings where custom devices are not available. This has been made possible thanks to a relatively sleek design, low price, simple adjustability, and (with regular cleaning) an achievable product life of 1 year.
In USA it may similarly provide a pragmatic therapy option for patients without access to custom devices or those needing an affordable complementary device for nights away from CPAP.
Patient selection
All prefabricated appliances have limitations and BluePro’s one size does not fit all. It will flex to fit most arch sizes but it can be difficult to achieve a good fit in patients with irregular occlusion, especially cross bites, large open bites or a significant curve of Spee. Patient selection and hands-on practice are therefore key to successful adoption in clinical practice.
For more information please contact hello@bluepro.pro or visit the website at www.bluepro.pro.
Case Study
 My passion is helping patients get a better night of sleep, which affords them a day void of nodding off at the wheel and a healthier relationship with their partner. However, with the constraints of medical insurance, the delivery of these custom-made devices is often delayed for months. This reality is not welcome by many patients, and without a quick and comfortable way to ameliorate the sleepiness and unwelcomed angst, they would leave my office discouraged.
My passion is helping patients get a better night of sleep, which affords them a day void of nodding off at the wheel and a healthier relationship with their partner. However, with the constraints of medical insurance, the delivery of these custom-made devices is often delayed for months. This reality is not welcome by many patients, and without a quick and comfortable way to ameliorate the sleepiness and unwelcomed angst, they would leave my office discouraged.
A typical example of this scenario is our patient Leah. She presented on Tuesday, desperate for a solution to her strained relationship with her fiancé. They were sleeping in separate beds, as the noise that accompanied her severe sleep apnea was not allowing him to sleep. As usual, the custom device would take a few months before delivery would be allowed by her insurance.
Enter BluePro. We have used many temporary appliances over the last 20 years, but my entire team loves the ease of fabrication, the unique adjustment mechanism and the fact they do not have to use any extra material. It appears to be the best complement to bridge the gap between custom and expediency.



