
by Ken Berley, DDS, JD, DABDSM
Standard of Care (SOC) is defined as: “What a reasonable and prudent practitioner would do in the same or similar circumstances.” SOC is a curious concept among medical professionals. Frequently, I discover that health care professionals do not understand that Standard of Care is a legal concept and is ultimately determined by a jury. Most professionals firmly believe that they determine the standard of care for their profession. To some degree that is correct. Health Care professionals (experts) present medical opinions in a court of law outlining what, in their opinion, should have been done to prevent a particular injury. Then a jury determines the standard to be applied. Until a jury rules on a particular therapy, no legal precedent or standard exists.
That begs the question; “Is the fabrication of a Morning Repositioning Device (MRD), within the SOC for the practice of Dental Sleep Medicine?” If not, should it be? Obviously, at this point no jury has determined that the inclusion of an MRD is legally mandatory, however, wouldn’t a reasonable and prudent practitioner fabricate an MRD to minimize the effects of mandibular advancement? In preparation for this article, I took an informal poll and found that only 52% of those polled routinely fabricated a MRD. When asked why MRDs were not routinely fabricated, I received excuses ranging from: not necessary; no reimbursement; don’t work, and patients don’t want another device.
In Dental side effects of mandibular advancement appliances – a 2-year follow-up.
J Orofac Orthop. 2008 Nov;69(6):437-47. doi: 10.1007/s00056-008-0811-9. Epub 2008 Nov 11. It was concluded that: Clinically small but statistically significant dental side effects predominantly affecting the incisors’ inclination occur after long-term wear of a Mandibular Advancement appliance. Overbite was highly significantly reduced (p = 0.006). The maxillary front teeth showed significant palatal tipping and those in the mandible significant labial tipping. Therefore, it is my opinion that tooth movement secondary to Mandibular Advancement may be the most significant long term complication of Oral Appliance Therapy. Therefore, from a risk management prospective, it is my legal opinion that a Morning Repositioning Device should routinely be employed to minimize this complication.
I have been unable to find any research which confirms that the use of a MRD will ultimately prevent tooth movement secondary to OSA, however clinically I see the results every day. Using a maxillary hard night-guard type MRD with an anterior ramp that is fabricated in centric occlusion will assist the patient in recapturing his/her centric bite each morning. The anterior ramp is inclined at a 60 degree angle and guides the mandible back into centric while simultaneously repositioning the lower anterior teeth into their original position. The hard acrylic night-guard will reposition the maxillary teeth functioning like a retainer or invisalign tray.
With the addition of a MRD to our treatment protocol, the issue of reimbursement always comes up. YES, I file for medical reimbursement for this device. I use the TMJ code S8262. This code is used whenever we are repositioning the mandible. I am well aware that we have traditionally used this code for TMJ therapy, however the code is not limited to that diagnosis. S8262 adequately describes the therapeutic function of a Morning Repositioning Device. Truthfully, only 50% +/- of my patients have medical benefits for S8262, however we routinely provide a MRD when treating OSA. If the patient does not have benefits for S8262, I use thermoplastic or one of the commercially available in-office techniques.
When filing medical insurance for S8262 with E0486 for OAT, you must be prepared to provide a letter of medical necessity for the S8262. I have included my letter for consideration.
To Whom It May Concern;
This is in response to your request for information regarding the treatment proposed for your insured, Mr. XXXX. Mr. XXXXX has been diagnosed with Obstructive Sleep Apnea, OSA 327.23. Mr. XXXX has been referred to our office by his sleep physician for treatment of his OSA with a Mandibular Advancement Device, MAD E0486. The treatment will include 2 different types of devices. The E0486 is a custom fabricated FDA approved Mandibular Advancement Device that will open his airway at night by advancing his mandible thereby controlling his OSA. By holding the mandible forward the airway is expanded and airflow is improved. The MAD maintains a patent airway which improves breathing and reduces OSA. Without this treatment Mr.XXXXX’s ____________ (co-morbid medical condition) can deteriorate which represents a major threat to his health.
The second device is S8262, a Mandibular Orthopedic Repositioning Device. This device is used in the morning to reposition the condyles in the glenoid fossa and to correct any dental misalignment that may have occurred during the night while wearing the MAD, E0486. The device is used to prevent TMD and tooth movement secondary to MAD therapy. Without this S8262 device, Mr. XXXXX could likely suffer TMJ Dysfunction and permanent displacement of the mandible in an anterior position, thereby causing permanent bite changes and necessitating orthodontic or surgical correction. This orthotic is medically necessary for the successful treatment of Mr. XXXXX’s OSA with a mandibular advancement device. The patient will be required to use both of these devices the remainder of his life unless surgical intervention or other treatment is instituted.
If you have any questions please feel free to contact me.
Sincerely,
Ken Berley, DDS, JD, DABDSM
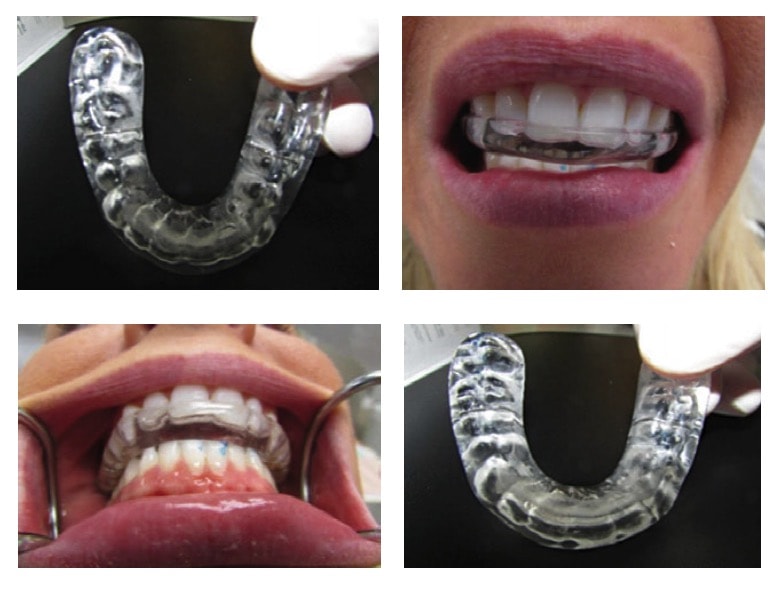 By including MRD in my treatment protocol, I have effectively increased my compensation for the treatment of OSA while simultaneously reducing the possibility of secondary tooth movement. This therapy provides a safeguard and an argument that you were proactively attempting to prevent any tooth movement. Coupled with a comprehensive informed consent, a MRD will effectively minimize your liability and would likely prevent any Malpractice action secondary to tooth movement.
By including MRD in my treatment protocol, I have effectively increased my compensation for the treatment of OSA while simultaneously reducing the possibility of secondary tooth movement. This therapy provides a safeguard and an argument that you were proactively attempting to prevent any tooth movement. Coupled with a comprehensive informed consent, a MRD will effectively minimize your liability and would likely prevent any Malpractice action secondary to tooth movement.
In Conclusion: Is MRD the Standard of Care for Oral Appliance Therapy? No; not at this time. However, in my professional legal opinion, with the research that we currently have, it is the only SMART way to treat OSA. If you are not already routinely constructing MRD’s for your patients with OSA, please help me make this the Standard of Care by adding this procedure to your protocol.
Stay Relevant With Dental Sleep Practice
Join our email list for CE courses and webinars, articles and more..

