A Commitment to Better OSA Therapy for All Patients
by Mark T. Murphy, DDS, FAGD, Lead Faculty for Clinical Education at ProSomnus Sleep Technologies and Len Liptak, MBA, CEO for ProSomnus Sleep Technologies

Not long ago a few dozen leading Dental Sleep Medicine clinicians convened to address a deceptively simple question: “Is it possible to engineer a better Herbst-style OAT device?”
Actually, the conversation commenced with a robust discussion on the feasibility of changing the coding for Medicare devices. The group opined that the current coding limited OAT device selection and diverged from AADSM guidance documents in certain areas. The ability to select from a variety of devices was seen as important for delivering effective and efficient OSA therapy to patients. Several clinical and industry leaders from many organizations have made tremendous efforts to change the coding – to no avail. If better OSA therapy for all patients is the objective, the reality of the current Medicare coding made it necessary to consider developing a next generation Herbst-style device. With a collective understanding, the group pivoted to the question of defining a better Herbst-style device.
Opportunities for a Better Herbst-style Device
The group advised that patients needed a Herbst-style device that was more comfortable. Currently available Herbst-style devices did an adequate job alleviating OSA. However, many patients had difficulty adapting to them. OSA is a life-long disease. Most patients were prescribed OAT after failing CPAP. It was important to have a Herbst-style device that patients could tolerate all night, every night. This guidance on comfort also extended to the desire to reduce the risk of side effects, such as the lower anterior crowding associated with traditional Herbst-style devices.
Importantly, this group of expert dentists explained that a Herbst-style device needed to be easy to use. Medicare patients are more likely to have compromised dentition. Considering the age and capabilities of general Medicare-eligible patients, the group listed several characteristics: The ideal device would be easy to keep clean. It should be easy to insert and remove. It should be more biocompatible. And easy to adjust, among other things.
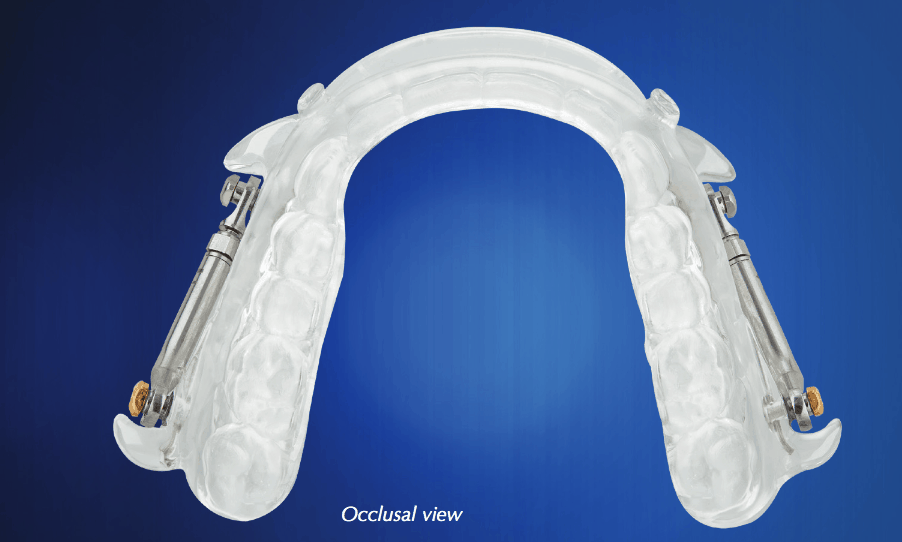 The group explained that they wanted a device that was efficient to prescribe, deliver and manage. Clinical efficiency was advanced as a tactic for offsetting reimbursement concerns. Medicare reimbursement rates were already low and had declined recently in some jurisdictions. They were not expected to increase any time soon. The count of Medicare beneficiaries is forecasted to increase by 30 million by 2030. Additionally, some private insurance companies have started adopting PDAC coding. Device breakage, slippage, discomfort, delamination and adjustments meant inefficiencies in the form of unplanned appointments. Moreover, Herbst-style devices were challenging to repair, expensive to replace if lost or damaged, and often meant the patient was without a device or placed in a temporary device during the repair or replacement process.
The group explained that they wanted a device that was efficient to prescribe, deliver and manage. Clinical efficiency was advanced as a tactic for offsetting reimbursement concerns. Medicare reimbursement rates were already low and had declined recently in some jurisdictions. They were not expected to increase any time soon. The count of Medicare beneficiaries is forecasted to increase by 30 million by 2030. Additionally, some private insurance companies have started adopting PDAC coding. Device breakage, slippage, discomfort, delamination and adjustments meant inefficiencies in the form of unplanned appointments. Moreover, Herbst-style devices were challenging to repair, expensive to replace if lost or damaged, and often meant the patient was without a device or placed in a temporary device during the repair or replacement process.
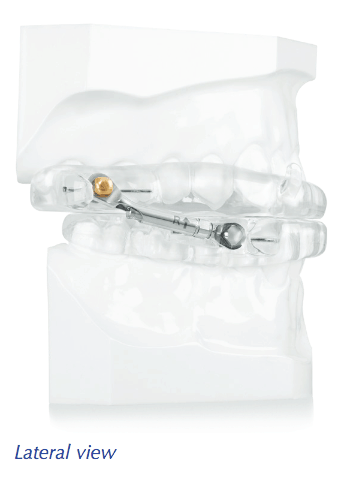 “The use of Herbst-style devices in Dentistry has been utilized in the dental world for many years. There has not been any change in the design of this appliance and that has left many of us in the field with few alternatives for treatment. The ProSomnus Herbst-style device development has taken the recommendations and frustrations of dentists and created a design that now respects the patient’s comfort, the doctor’s desires and the overall improvement in compliance and functionality that is expected in a superior product. Kudos to ProSomnus for the vision to make things better for the doctors and our patients that trust us so well.” – David B. Schwartz, DDS, D-ABDSM
“The use of Herbst-style devices in Dentistry has been utilized in the dental world for many years. There has not been any change in the design of this appliance and that has left many of us in the field with few alternatives for treatment. The ProSomnus Herbst-style device development has taken the recommendations and frustrations of dentists and created a design that now respects the patient’s comfort, the doctor’s desires and the overall improvement in compliance and functionality that is expected in a superior product. Kudos to ProSomnus for the vision to make things better for the doctors and our patients that trust us so well.” – David B. Schwartz, DDS, D-ABDSM
This is the spirit of collaboration that gave birth to the First Precision Herbst-style OAT Device, the ProSomnus® [PH]. The ProSomnus [PH] is an FDA cleared, PDAC approved sleep device. This exceptional group of dentists wanted a better OAT experience for their Medicare patients and other patients who might benefit from a Herbst-style device. But how, within the PDAC constraints, does one engineer a better Herbst-style Device?
The ProSomnus precision OAT platform would form the foundation for the ProSomnus [PH] Precision Herbst-style device. The ProSomnus precision platform is a unique confluence of forward engineering, precision manufacturing and material science that enables the Company to create innovative OAT devices with meaningful advantages. It is the same precision OAT platform that enables the ProSomnus [IA] and [CA] devices that have been used to successfully treat tens of thousands of patients by many leading Dental Sleep Medicine clinicians, reinforced by clinical studies demonstrating efficacy, effectiveness, adherence and mitigation of side effects like tooth movement.
Developing ProSomnus [PH]
Precision means confidence that the prescription is faithfully and accurately represented in the device design such that the clinician will spend more time treating the patient and less time compensating for the device (i.e. making up for handmade human error). Precision means responsiveness to the subtleties of patient phenotypes while minimizing variances in the production of a Medical Device. Precision means using materials that are stronger, more biocompatible, and enable optimized device designs. Precision means simulating titrations when designing devices to ensure symmetry and design harmony throughout treatment. In sum, precision means the opportunity for dentists to create a better patient and practice experience.
“I think the ProSomnus [PH] is the best Herbst-style appliance on the market to date. The ProSomnus platform enables a very slim profile which is a plus, also the added bumper feature helps patients so they won’t feel the screw mechanism. My patients love the smaller, smoother, new design and protection of their cheeks. Patients can easily advance the nut with the tool provided. I love providing my patients with a better oral appliance option. Makes my job easier too, which is a win-win.”– Erika Mason, DDS, D-ABDSM, D-ACSDD
A More Comfortable Choice
Patients and clinicians have noted that, “It is smaller.” This is not an accident. It is by design. The ProSomnus [PH] device has 18% less overall volume (fig. 1), on average. It also has 83% less lingual splint thickness (fig. 2), for the purpose of optimizing protrusiveness and comfort while minimizing the risk of side effects.
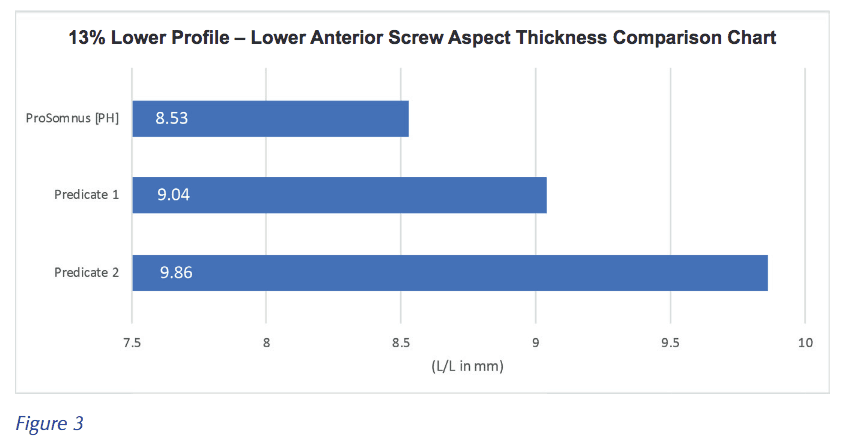
Patients have also noted that it is, “Much more comfortable.” Again, this is by design. ProSomnus [PH] has specific design characteristics that are intended to make the device more comfortable. The ProSomnus [PH] features comfort bumps that are intended to protect the lip commissures and cheeks. Comfort bumps come standard with the ProSomnus [PH]. The comfort bumps can be easily removed, for example, if there are concerns of interference with the coronoid process. ProSomnus [PH] also has a 13% lower profile (fig. 3) in the anterior screw aspect, than traditional Herbst-style devices. The attachment screws are capped for comfort and hygiene.
“We had become accustomed to our patients complaining as soon as they were shown a Herbst-style design. This all changed when the ProSomnus [PH] became available, as the transparent design along with the shielded hinges are much less off-putting to our patients.” – B. Kent Smith, DDS, D-ABDSM, D-ASBA
An Easy Choice
 Patients who have switched from a traditional Herbst remarked that it is, “Way easier to use.” The ProSomnus [PH] offers several features that are designed to make the device easier for patients and team members. The upper arch features a gold color screw for orientation during insertion. The hex nut titration screw is designed to be more definite, minimizing slippage. The ProSomnus [PH] features metal-free hooks for elastics.
Patients who have switched from a traditional Herbst remarked that it is, “Way easier to use.” The ProSomnus [PH] offers several features that are designed to make the device easier for patients and team members. The upper arch features a gold color screw for orientation during insertion. The hex nut titration screw is designed to be more definite, minimizing slippage. The ProSomnus [PH] features metal-free hooks for elastics.
“ProSomnus listened and has created unique changes to the traditional Herbst appliance. One of which is the creation of ‘comfort bumps’. Now the most common complaint from patients using a Herbst-style appliance has been successfully addressed!” – Kenneth A. Mogell, DMD, D-ABDSM
A More Efficient Choice
The ProSomnus [PH] features the most flexible titration range of any Herbst-style device. It comes with a standard 7.0mm titration range, 6.0mm of forward range and 1.0mm of backward range. And as with all ProSomnus devices, additional titration arches can be ordered if more advancement range is needed.
The ProSomnus [PH] features a unique, best in class, 3+2 Year Warranty. The [PH] comes standard with a 3-year warranty, plus a free-of-charge option to add 2 more years for Medicare patients. Additionally, the ProSomnus [PH] is designed to be easy to repair and replace. Herbst-style arms are easy to replace if needed. The device can be precisely replicated from existing records if there is a need for a replacement. The patient can be converted to a ProSomnus [IA] or [CA] device from existing records if there is a need, or if the patient simply desires a backup device.
Like so many leading Dental Sleep Medicine clinicians, the commitment to better OSA therapy compels ProSomnus Sleep Technologies to continuously advance. Identify. Analyze. Innovate. That is the basic mantra of continuous improvement. Though ProSomnus manufactures precision OAT devices, it isn’t about the device. It is about treating the patient with OSA. It is about developing and manufacturing devices that can make it easier for the dental team, in small but meaningful ways, to create meaningful and successful treatment experiences.
 It is a commitment and passion, supported by technical excellence and expert clinical insights, that led to the creation of the ProSomnus [PH] Precision Herbst-style Sleep and Snore Device – a new option for helping clinicians deliver the best possible OSA therapy for their patients.
It is a commitment and passion, supported by technical excellence and expert clinical insights, that led to the creation of the ProSomnus [PH] Precision Herbst-style Sleep and Snore Device – a new option for helping clinicians deliver the best possible OSA therapy for their patients.
Read more about how the ProSomnus [PH] Herbst-style device and how it now qualifies for Medicare reimbursement!




